Health Information Technology Proposed Rule (HTI-2) is Out
ONC will be hosting information sessions in the coming weeks, including an overview session on July 17 at 2:00 PM ET.
Global Internet Outage Continues to Impact Many Healthcare Systems
Human error may be just as much of a risk as hacking, ransomware and other intentionally malicious assaults.
2025 Medicare Physician Fee Schedule is Out (and CMS Did Not “Package Price” Cellular Tissue Products / Skin Substitutes in the Doctor’s Office)
The billing of CTPs in the doctor’s office is unchanged. The 2025 Medicare conversion factor is set to decrease for the fifth straight year.
Breaking News! OPPS Proposed Rule is Out!
At first glance, there are no changes to Cellular Tissue Product /skin substitute payment, which remain under package pricing in this site of care.
Accelerated and Advance Payment (AAP) for Medicare Ends on July 12
CMS announced that Medicare payments under the Accelerated and Advance Payment (AAP) Program for the Change Healthcare/Optum Payment Disruption (CHOPD) will end on July 12.
CMS Adds 76 New Items to its Master List of DME Supplies That May Be Subject to Prior Authorization (and Some Surgical Dressings are on the List)
The Centers for Medicare & Medicaid Services (CMS) added 76 new items and deleted three from its master list of durable medical equipment (DME), prosthetics, orthotics, and supplies.
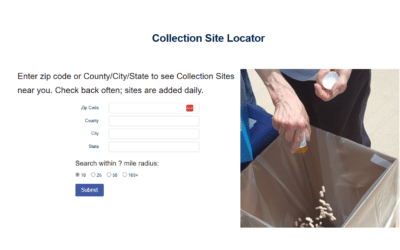
How the DEA is Working to Help Communities Get Rid of Unused or Expired Prescription Medications
You can find a medication collection site by putting in your zip code on the Collection Site Locator.
How to Subscribe to the NGS Self Service Plus Newsletter
If NGS is your MAC, you might want to subscribe to the Self-Service Plus newsletter, accessed online.
Physicians Have Until Dec. 31 to File a MIPS Hardship Exemption Due to the Change Cyberattack
The 2024 MIPS EUC portal is now open, and physicians have until Dec. 31 to file a hardship application and avoid a 2026 MIPS negative payment adjustment.
Skin Substitute Proposals: What You Can Do
These proposals are in a comment period until June 8, 2024. Any interested stakeholder may submit comments to share their feedback on these proposals.
What Doctors Wish Patients Knew About Prior Authorization
Maybe if I’d let patients read this article, I could have discussed the issue with my patients honestly, rather than just running away from it.
Pneumatic Compression Devices Are in the Spotlight for 2024 Audits
Check out the article by Knicole Emanuel in Today’s Wound Clinic: Pneumatic Compression Devices Are in the Spotlight for 2024 Audits.
CMS Explains its 2024 Value-Based Care Strategy
CMS ALWAYS tell us what they are going to do before they do it. Then they do it, and we are surprised.
A Little Reprieve for Physician Pay Cuts
A 3.4% Medicare physician pay cut took effect on January 1, 2024. However, thanks to the passage...
MIPS Quality Measure #130 Documentation
Too many providers are under the impression that merely documenting the patient’s medication list satisfies the performance of this measure, and it does not.
CMS Announces Flexibilities in the Wake of the Change Cyberattack
CMS is encouraging MA plans to offer advance funding to health care professionals most affected by the cyberattack.
CMS Announces Reopening of 2023 MIPS EUC Application in Response to Change Healthcare Cyberattack
I am reposting the entire message from the CMS about the extension of the Extreme and Unavoidable...
CMS Removes the Requirement for AUC From the 2024 Medicare Physician Fee Schedule
Implementing the AUC program would have been a time consuming and expensive lift for practitioners.
Update on the Surgical Dressing Policy – Thanks to the Alliance of Wound Care Stakeholders
The Alliance of Wound Care Stakeholders has had another policy win!
G2211 Documentation
Last month’s blog explained HCPCS G2211, its appropriate use, and inappropriate use. HCPCS G2211 -...
Resources from CMS and the AMA to Help You Implement Changes to Physician Documentation & Billing
There have been two major changes in the regulations pertaining to physician documentation during ambulatory visits, both of which are favorable for wound care practitioners.
Medicare Audits on Pneumatic Compression & Surgical Dressings – “It’s About the Documentation…”
I am a huge fan of attorney Knicole Emanuel’s blog and her most recent post is directly relevant to the field of wound care.
Be Careful Billing the New CMS G2211 Add-on Code for Visit Complexity
It is intended to “better recognize the resource costs associated with evaluation and management visits for primary care and longitudinal care.”
Claims Disruptions Continue After Cyberattack on Change Healthcare
This is yet another blow to hospitals and physicians that are struggling to make ends meet in the face of payment reductions.
Ransomeware Cyberattack on Change Healthcare is Affecting Individual Physician Practices Too
United Health-owned Change Healthcare has confirmed the ransomware group “ALPHV/Blackcat” is...
Noridian Has Begun Requesting “Documentation to Support Credentialing of the Provider Performing the HBO Service”
If there are super-secret requirements that Noridian has now decided to impose, then they should be enshrined in a public document somewhere.
New Coding When Caring For a Complex Condition
G2211 should be added to an office or other outpatient evaluation and management CPT® code. It should never be submitted alone.
CMS Final Rule on Prior Authorization Released
Prior authorization (PA) is costly, inefficient and responsible for patient care delays. The...
Surgical Dressing Webinar by Noridian on March 7th
On Thursday, Mar 7, 2024 from 12:00 PM – 1:00 PM CST, Noridian is offering a webinar on Surgical Dressings. It is intended for DME suppliers.
Noridian Offering Tutorials on Hyperbaric Oxygen Therapy (HBOT)
Noridian is offering two self-paced training tutorials to “assist providers and facilities in better understanding Hyperbaric Oxygen (HBO) Therapy.”
Novitas Releases Top Ten Reasons for Claims Denials…and Wound Centers Should Especially Check Out Reason #7
Reason #7 for claim denials is especially relevant to hospital-based outpatient wound centers (HOPDs).
MIPS Revenge!
The MIPS program has continued to evolve with penalties increasing and the participation threshold needed to avoid a penalty also increasing.
Noridian is Offering a Feb 13th Webinar on the Lymphedema Compression Benefit Category
On February 13th, 2024, Noridian is offering a Webinar for DME suppliers about lymphedema compression benefits.
Pneumatic Compression Devices Webinar by Noridian on Feb 13th, 2024
On Tuesday, Feb 13, 2024 from 2:00 PM – 3:00 PM CST, Noridian is offering a webinar intended for DME suppliers about pneumatic compression devices (PCDs).
The Deadline to Submit MIPS Data for 2023 is April 1 at 6 pm EST to Avoid up to a 9% Penalty on Medicare Revenue
The data submission period for Medicare’s Merit-Based Incentive Payment System (MIPS) 2023 performance year is underway.
Time Sensitive! It’s Not Too Late to Sign the Letter to CMS Protesting the 41.5% Reduction of HBOT Facility Fees!
I know the holidays are busy – but most of us would like to have a job to go back to in January…
CPT® Change for 2024
There are major changes to CPT coding associated with noncontact near-infrared spectroscopy.
Without Warning – the Outpatient Prospective Payment Final Rule Cuts HBOT Facility Fees by 40%
What we need is for people to submit a comment, and to spread the word that hyperbaric oxygen therapy is in a crisis.
ICD-10 Changes
A new ICD-10 Clinical Modification (ICD-10-CM) code set took effect October 1, 2023. In this new code set, there are many code additions, deletions, and modifications.
Modifications Needed to Wound Center EHR to Support DME Orders for Dressings
Your EHR will need to be modified to meet the DME documentation requirement burden (and if you think the list below is ridiculous – it is, but it’s still required).
Mindfulness, Electronic Health Records and Ebola
A recent Ebola outbreak in Dallas was blamed on a “flaw” in the hospital’s electronic health record (EHR). Yes, you heard me correctly.
JZ Modifier Use With Skin Substitutes
CMS has issued guidance requiring the use of Modifier JW on all claims for biologicals from single-dose containers that are separately payable under Medicare Part B when there are unused / discarded amounts.
Observations About the 2024 PFS Pertaining to Cellular and/or Tissue-Based Products in the Doctor’s Office Setting
On July 13, 2023, CMS issued proposed rules for the 2024 Physician Fee Schedule (PFS) and the Hospital Outpatient Prospective Payment System (OPPS).
“Hidden RAC Audits” – Seriously…
Attorney Knicole Emanuel has posted a chilling blog on her site about hidden RAC (Recovery Audit Contractor) audits.
Ulcer Debridement & Total Contact Cast Application
We cannot submit CPT®[1] coding for both ulcer debridement and total contact cast application when both are performed for the same ulcer at the same encounter.
Listen to a Podcast with Dr. Helen Gelly, “How to Get Your Wound Clinic Through an Audit”
On your drive home, check out this podcast with Helen Gelly MD, FUHM, FACCWS, UHM/ABPM, “How to Get Your Wound Clinic Through an Audit.”
HBOT Facility Payments are Under Attack (Physician Supervision is Next…)
We need individuals at all training levels to comment on the proposed changes that CMS is proposing in the Medicare Physician Fee Schedule (MPFS) for the technical payment of HBOT.
Check Out the Long List of CTPs (“Skin Subs”) Not Covered in the CGS, First Coast, & Novitas LCDs
Three Medicare Administrative Contractors (MACs) issued their final Local Coverage Determinations (LCDs) for Cellular and/or tissue-based products (CTPs).
Highlights of the 2024 Outpatient Prospective Payment System (OPPS)
You should read the documents yourself, but here are some highlights of the 2024 OPPS proposed rule (in no particular order).
PRESS RELEASE: New Study in Journal of Medical Economics Shows Decrease in Chronic Wound Medicare Costs Amid Increase in Chronic Wound Prevalence
Over 2014-2019, the number of Medicare beneficiaries with chronic wounds increased 13% to 10.5 million amid a surprising 20% reduction in chronic wound expenditures. Researchers seek to explain why, and what the policy implications may be.